Engineering Chemistry 1 important 2 marks
UNIT – I Water Technology
1. Define hard water and soft water.
Ans:Water which does not produce lather with soap solution but forms a white precipitate is called hard water. Water which lathers easily with soap solution is called soft water.
2. Give a chemical test to detect hardness of water.
Ans:Hardness can be detected by treating water with soap, as hard water forms a white precipitate with soap solution.
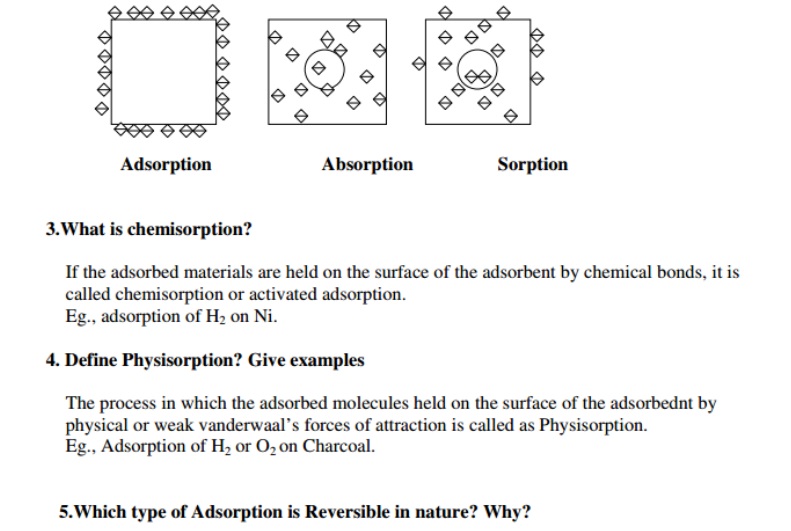
3. What is aeration? Why is it required?
Ans:Aeration is a process in which water is mixed with air.
The main purpose of aeration is-
1. To remove CO2, H2O and other volatile substance causing bad taste and odour.
2. To remove impurities like Fe and Mn as their respective hydroxide.
4. Distinguish between priming and foaming.
Ans:Priming is the process of production of wet steam during the process of steam production in boilers. During steam production certain water droplets also get into steam due to some dissolved solids in water, high steam velocity or irregular steam velocity. Foaming is the production of persistent bubbles in the water which do not break easily due presence of oily or soapy substances.
5. What are boiler compounds? Give examples.
Ans:The chemicals added inside the boilers (during internal treatment of water) to remove scale forming substance are called boiler compounds. Eg. Sodium phosohate, calgon.
6. What is calgon? What is its role in water treatment?
Ans:It is chemical added inside the boilers during internal treatment of water. Calgon is sodium hexametaphosphate. (NaPO3)6 or Na2[Na4(PO3)6 It forms a highly soluble complex with Ca2+ ions of hard water and prevents the formation of scale in boilers.
7. What is desalination? Name few methods of converting sea water into fresh water?
Ans:The process of removal of extra common salt from sea water is described as desalination or desalting.
The various desalination processes
1. distillation
2. freezing
3. reverse osmosis
4. electro dialysis
8. What is meant by break point chlorination?
Ans:Chlorination of highly polluted water such that after all the bacteria are killed, the organic impurities present in water are completely oxidized and the residual chlorine in water is about 0.1 ppm is called break point chlorination.
9. Define Temporary and permanent Hardness.
Ans:The hardness removed by boiling the water is called as temporary hardness and it is due to the presence of carbonate and bicarbonate of calcium and magnesium. The hardness due to the presence of sulphate and chlorides of calcium and Magnesium is called as permanent Hardness.
10. Name any two disadvantages of using hard water for domestic purpose.
Ans:
1. Hard water when used for drinking affects our digestive system and also leads to the formation of stones in kidney.
2. Hard water when used for washing does not lather with soap easily and thereby causing wastage of soap.
11. Discuss ozone gas treatment.
Ans:Ozone is an unstable allotrope of oxygen. It is a powerful disinfectant. Ozone is readily absorbed by water. Ozone contains three oxygen atoms and one atom breaks easily. This produces nascent oxygen [O] which is a powerful disinfectant. It kills all bacteria and does not produce any odour or taste.
12. Explain ultraviolet treatment. Ultraviolet rays are powerful disinfectant.
Ans:They are produced by passing electric current through mercury vapour lamp. When water is allowed to flow in thin stream. UV rays from the lamp destroys pathogens completely.
13. Define nuclear fission reaction.
Ans:It is defined as the process of splitting of heavier nucleus into two or more smaller nuclei by bombarding with slow speed neutrons with simultaneous liberation of large amount energy.
14. What are batteries? Mention the basis for their functioning.
Ans:`Batteries may be considered as store houses for electrical energy on demand. The basis for battery technology is that the chemical energy derived from the chemical reactions in the battery is converted into electrical energy.
15. Explain alkaline battery.
Ans:This is an improved form of dry cell. Here, Anode: Zinc, Cathode: MnO2, Electrolyte: KOH This cell drives power from the reduction of MnO2 cathode and the oxidation of Zn anode. The emf of the cell is 1.5V. It is used in calculators, watches etc.
16. What is a storage cell?
Ans:A storage cell is the one which can operate both as a voltaic cell and as an electrolytic cell. When it acts as a voltaic cell, it supplies electrical energy and becomes run down. When it is recharged, the cell operates as an electrolytic cell.
17. Write few applications of solar cells.
Ans:a. Solar cells are used in boilers to produce hot water for domestic and industrial uses. b. Solar cells can be used for lighting purposes. c. Solar cells can be used to drive vehicles. d. They can be used to produce H2 by electrolysis of water.
18. A sample of water contains 120mgs of MgSO4 per litre. Calculate hardness in terms of CaCO3 equivalents.
Ans:Given : The amount of MgSO4 = 120 mgs/lit. Amount equivalent to = The amount of hardness producing salt x 100 CaCO3 Molecular weight of hardness producing salt We know that, the molecular weight of MgSO4 = 120 Therefore, amount equivalent to CaCO3 = 120 x100 120 = 100 mgs/lit.
19. Write the uses of Lead Storage battery.
Ans:
(i) It is used in the automobiles.
(ii) It is used in gas engine ignition, telephone exchanges, Hospitals, power stations.
20 . What is meant by boiler corrosion? How is it prevented?
Ans:Boiler corrosion is due to the presence of dissolved gases such as O2 CO2 and easily hydrolysable salts t like MgCl2. Mechanical deaeration is a method used to remove dissolved gases such as O2 and CO2. The principles involved in this process are the solubility of a gas is directly proportional to pressure and inversely proportional to temperature.
21. What are the requisites of drinking and boiler feed water? (Nov 2001)
Ans:Boiler water must be free from hardness causing substances, dissolved gases, oil, silica. Water should be clean, colourless and odourless. It should be free from pathogenic organisms. It should not be excessively hard or soft.
22.What is a primary battery? Give an example (June 2006) (Or) What are primary cells?
Ans:In primary cells, the electrode and the electrode reactions cannot be reversed by passing an external electrical energy. The rections occur only once and after use they become dead. Therefore, they are not chargeable. Eg Dry cells.
23. How are anodic and cathodic electroactive materials made in Ni-Cd battery?
Ans:Nickel-cadmium cell consists of a cadmium anode and a metal grid containing a paste of NiO2 acting as a cathode. The electrolyte in this cell is KOH.
For further syllabus, notes, video materials and question papers visit kprblog.in
Join us on facebook @ KPR BLOG
Mail your Feedback/Queries to [email protected]